 Abbott, the global healthcare company, today announced that its FreeStyle Libre 3 continuous glucose monitoring (CGM) system, the latest addition to the world-leading FreeStyle Libre sensor technology, is now available for use in the UK.
Abbott, the global healthcare company, today announced that its FreeStyle Libre 3 continuous glucose monitoring (CGM) system, the latest addition to the world-leading FreeStyle Libre sensor technology, is now available for use in the UK.
The FreeStyle Libre 3 system is available via the NHS Supply Chain Framework for people with more complex diabetes management needs who require a real-time CGM device. For example, those with impaired hypoglycaemia awareness.
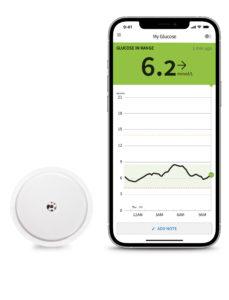
The FreeStyle Libre 3 system automatically delivers continuous, real-time glucose readings every minute to users’ smartphones2 and provides 14 days of unsurpassed accuracy4 with the smallest and thinnest1 sensor design available. As a result, glucose monitoring with Abbott’s FreeStyle Libre 3 system is even more convenient and discreet3.
The sensor, which is only the size of a £1 coin, is worn on the back of the upper arm for up to 14 days. Reducing the system’s total volume by more than 70%,[i] Abbott designed the FreeStyle Libre 3 system to be more sustainable for the environment, including a 41% reduction in plastic and 43% decrease in carton paper.5
The system comprises the sensor and the FreeStyle Libre 3 smartphone app2, which allows users to record their real-time glucose levels, glucose history and trend arrows and see them every minute. A quick glance at their compatible smartphone is all it takes to see changes in glucose levels and adjust their diabetes management accordingly.
“At Abbott, we are proud to bring the FreeStyle Libre 3 system to the UK for people with diabetes. For those people with more complex diabetes management needs, the system aims to make management easier and more convenient by providing a continuous picture of their glucose levels in real-time, as well as where they are heading,” explained Neil Harris, General Manager, Abbott’s UK and Ireland diabetes care business.
Eligibility criteria for real-time CGM can be found on the Diabetes UK or JDRF websites. Those who think they may be eligible should discuss with their healthcare professional at their next routine appointment.
The FreeStyle Libre 3 app is initially available for Android operating systems only, with the iOS version expected in the coming months.
Abbott’s FreeStyle Libre and FreeStyle Libre 2 systems remain listed on the NHS Drug Tariff and available on prescription for people with diabetes eligible for Flash Glucose Monitoring.
Abbott’s FreeStyle Libre system is designed to change the way glucose is monitored in people with diabetes and ultimately help them improve their diabetes management[ii]. The system measures glucose levels using a sensor worn on the back of the upper arm, eliminating the need for routine finger-pricking[iii].
Three FreeStyle Libre systems; FreeStyle Libre, FreeStyle Libre 2 and FreeStyle Libre 3 are now available in the UK, offering a range of glucose monitoring solutions depending on individual need.
Numerous clinical studies have shown that people who use the FreeStyle Libre systems experienced improved glucose control[iv], increased time in target range[v], reduced hyperglycaemia (high blood glucose) and hypoglycaemia (low blood glucose) [vi], and lower HbA1c (average glucose concentration over 3 months)11 – all of which contribute to improving the health of people with diabetes. Data also show that use of the FreeStyle Libre system reduces diabetes-related hospitalisations and workplace absenteeism11. These aspects also promote an increase in quality of life.[vii]
As the most widely used sensor-based glucose monitoring system in the world[viii], Abbott’s FreeStyle Libre product portfolio has changed the lives of nearly 4 million people in over 50 countries around the world.
[i] Data on file, Abbott Diabetes Care, Inc.
[ii] Bolinder, Jan, et al. Novel glucose-sensing technology and hypoglycaemia in Type 1 diabetes: a multi-center, non-masked, randomized, controlled trial. The Lancet 388.10057 (2016): 2254-2263.
[iii] Additional testing of glucose levels using a blood glucose meter is required if symptoms are not consistent with system readings or alarms.
[iv] Eeg-Olofsson et al. Sustainable HbA1c decrease at 12 months for adults with Type 1 and Type 2 Diabetes using the FreeStyle Libre System: a study within the National Diabetes Register in Sweden.
[v] Lang J, Jangam SR, Dunn TC, Hayter G. Expanded real-world use confirms strong association between frequency of flash glucose monitoring and glucose control.
[vi] Dunn TC, Xu Y, Hayter G, Ajjan RA. Real-world flash glucose monitoring patterns and associations between self-monitoring frequency and glycaemic measures: a European analysis of over 60 million glucose tests. Diabetes Res Clin Pract. 2018;137:37-46.
[vii] Fokkert M, van Dijk P, Edens M, et al. Improved well-being and decreased disease burden after 1-year use of flash glucose monitoring (FLARE-NL4). BMJ Open Diab Res Care. 2019;7(1):
e000809. doi:10.1136/bmjdrc-2019-000809.
[viii]Data based on the number of users worldwide for the FreeStyle Libre system compared to the number of users for other leading personal use, sensor-based glucose monitoring systems. Data on file, Abbott Diabetes Care, Inc.



