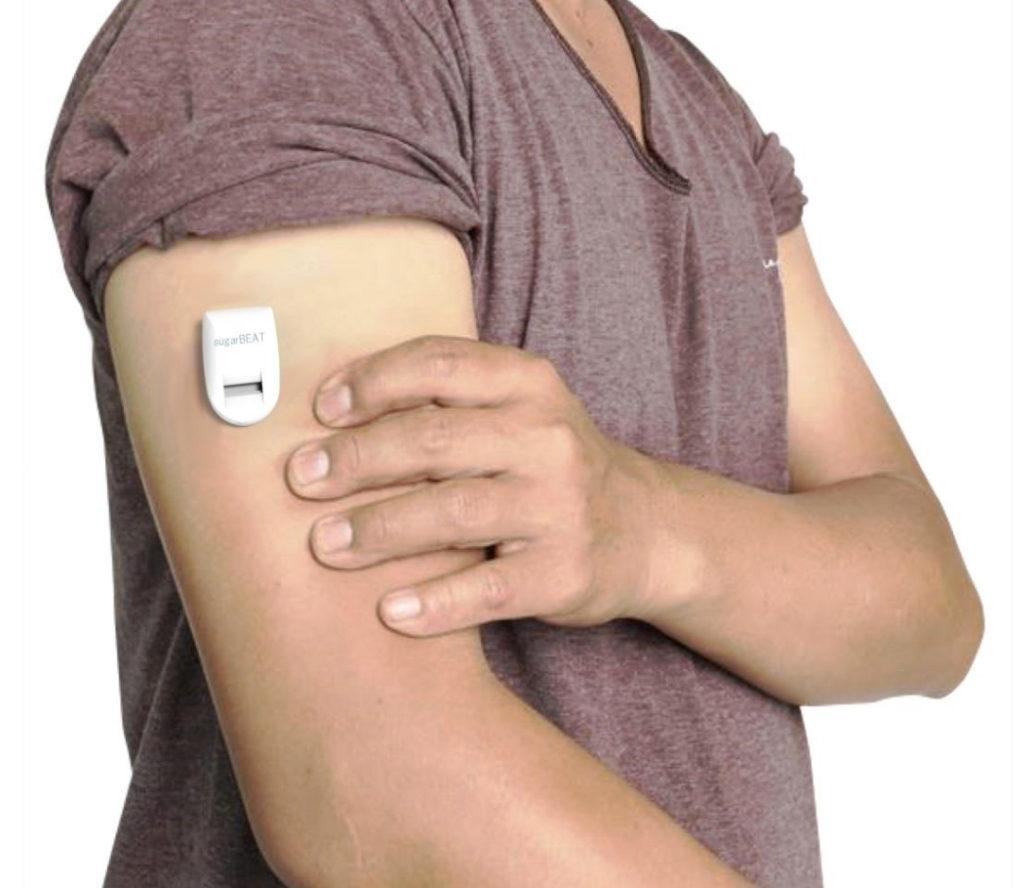
Nemaura Medical is a medical technology company focused on the development of SugarBEAT as a non-invasive, affordable and flexible Continuous Glucose Monitor (CGM) for use by people with diabetes (PWDs) and pre-diabetes, has submitted its medical device application to the U.S. Food & Drug Administration (FDA) for the device following the successful completion of clinical studies and two summative human factors usability studies for it.
The clinical studies were split between Type I and Type II PWDs, and consisted of 75 patients over 225 patient days. The studies generated over 12,000 paired data points, with blood samples taken via catheter every 15 minutes over a 12-hour period for three non-consecutive days for each patient. The study design was based on two previous pre-sub meetings Nemaura held with the FDA.
Dr. Faz Chowdhury, CEO of Nemaura based in Loughborough UK, comments, “Currently, CGM usage among pre-diabetics and other non-insulin using diabetics is negligible. Unlike other devices on the market, as the world’s first non-invasive CGM, SugarBEAT does not puncture the skin to insert a sensor filament inside the skin. Instead the sensor sits on top of the surface of the skin. For this reason, Nemaura believes its market to be many times larger than the market addressable by the current competition. The FDA has a structured review process, which should allow the review to be expedited, and while awaiting feedback on the application, the Company has now started to enter into discussions with key global companies in this space for commercialization in the US.”
Insulin users can adjunctively use SugarBEAT when calibrated by a finger stick reading. SugarBEAT consists of a daily, disposable adhesive skin-patch connected to a small form factor rechargeable transmitter, connected via Bluetooth to a specially designed mobile application, which displays glucose readings at five-minute intervals throughout the day. www.SugarBEAT.c