In this article I look at some of the clinical trials being undertaken in what is being refer to as a ‘bionic’ or ‘artificial’ pancreas (AP), also known as close-loop insulin delivery. It is an overview and therefore not entirely comprehensive. As normal, further updates will be included as news reports or further features in subsequent issues of this magazine as this is not a static field, progress is happening all the time, if slowly. — Sue Marshall
In March 2012, the American Federal Drugs Administration (FDA) allowed the first outpatient clinical trial of an artificial pancreas (AP) system. Five years later there are several hundred sites around the world conducting artificial pancreas clinical trials — many of them in outpatient settings (so at home, not confined to a hospital or clinic).
The purpose of trying to produce an artificial pancreas is two-fold. First, it’s to improve overall blood glucose control, whether that is judge by HbA1c or time-in-range (another way of assessing glucose control that is gaining credibility as a better guide to good control than a number that is just a three-month average). Second, it’s to improve quality of life, creating systems that can take away some of the burden of living with diabetes. If CGM is accurate, if we trust the pumps and the algorithms within them to work properly, then certainly there is growing proof that closed loop improves outcomes.
Closed loop means letting a CGM sensor assess blood glucose levels, beam them into an associated insulin infusion pump, and allow the algorithm in the pump to decide on what to do next in terms of insulin delivery. That means that the person with diabetes is not deciding whether to increase the insulin delivery or suspend it, the equipment is. A fully automated system will sense rising glucose levels, including at mealtimes, and adjust insulin automatically.
Various ways of delivering insulin.
Anyone interested – diabetics themselves, their partners parents or carers, the healthcare community as well as the manufacturers of these technologies — need to assess the risks involved with an automated system that delivers insulin without human intervention. Clinical trials do this usually in a dedicated hospital environment, though that is just to be on the safe side. In reality, there’s little point in any of these systems if they won’t work in a real-world setting, so it’s good to know that now trials are increasingly working around participants being in their normal home (and work) environments.
The ‘risk’ probably sounds more dramatic than it is. Anyone taking insulin has to make a serious medical decision every time they dose, whether by an insulin shot or an insulin pump bolus and risk having hypos as a result. For us, its business as usual, we’d just be using a different bit of kit.
In development
Last year, the Journal of Diabetes Science and Technology published a report, “Artificial Pancreas Device Systems for the Closed-Loop Control of Type 1 Diabetes, What Systems Are in Development?” which was overseen by Sara Trevitt (BSc, PhD, NIHR Institute of Applied Health Research, University of Birmingham). The report look at closed-loop artificial pancreas device (APD) systems, which it define as being externally worn medical devices that enable people with Type 1 diabetes to regulate their blood glucose levels in a more automate way. It state that, “The innovative concept of this emerging technology is that hands-free, continuous, glycaemic control can be achieved by using digital communication technology and advanced computer algorithms.”
A ‘horizon scanning’ review of this field was conducted using online sources to identify systems in development. The systems were classified into subtypes according to their level of automation, the hormonal and glycaemic control approaches used, and their research setting. The result was that 18 closed-loop APD systems were identified. All were being tested in clinical trials prior to potential commercialization. It was estimated that three systems could become commercially available in the EU during 2017
As the reliability and use of Continuous Glucose Monitoring (CGM) continues to increase, then an artificial pancreas starts to look like a reality, combining CGM with an insulin pump and an algorithm that automates the control and suspension of insulin infusion.
The combination of CGM and insulin pump has been around a few years now, but only last year a pump came out that was ‘sensor-augmented’. Medtronic’s 640g combines insulin infusion, CGM and SmartGuard technology. This allows the pump to suspend insulin delivery if CGM glucose readings are low. Available in the US and UK, one of the main upsides of this pump, CGM and ‘low glucose suspend’ is to virtually eliminate night-time hypos and also leads to improved overall HbA1c results. Though often modest, such improvements are known to reduce the risk of long-term complications as a result of living with diabetes.
Then in November 2016, Medtronic’s latest version of it’s MiniMed pump series, the 670G hybrid closed-loop system, was approved by the FDA and was hailed at the time as being the first AP to have been approved. It is intended for people aged 14 years or older who have Type 1 diabetes. Because it operates with a smart algorithm that learns an individual’s insulin needs and delivers appropriate basal doses 24 hours a day, little user input is required. Diabetics who use the system will only have to enter their mealtime carbohydrates, accept bolus correction recommendations, and periodically calibrate the sensor. Not yet available in the UK, the fact that it’s passed a major milestone (getting approved by the FDA) means that it’s on its way. In this case hybrid refers to the ability of the pump to both suspend as well as to increase insulin delivery.
In the loop

The bionic pancreas system developed by Boston University (BU) .
As well as low-glucose suspend, it is possible to automate pumps to automatically increase basal rates or even give an insulin bolus. These however are not yet available. Closing the loop will be taking out human intervention, letting the algorithms in the pumps decide on whether or not to suspend insulin delivery or increase the basal level of insulin delivery or give a corrective bolus. Some models are incorporating two pumps – one to deliver insulin, the other to deliver glucagon in case of a hypo.
Note that research into closed loop or AP is going on globally, with much input from JDRF in the US and Diabetes UK in Britain. These are some trials taking place currently.
In the US, Tandem Diabetes Care and TypeZero Technologies have accelerated development and commercialization of a Closed-Loop Artificial Pancreas System. In July 2016 Tande, a medical device company and manufacturer of a family of innovative touchscreen insulin pumps, and TypeZero, a digital health and personalized medicine company, entered into a License Agreement allowing Tandem to integrate TypeZero’s AP technology into a next-generation insulin pump.
TypeZero’s AP technology includes a series of treat-to-target algorithms developed by TypeZero from initial research conducted at the University of Virginia. To date, this technology has been used in more than 28 clinical studies involving more than 475 participants, with data referenced in a number of journal articles.
Bigfoot Biomedical
Bigfoot Biomedical was founded by people affected by and living with Type 1 Diabetes (T1D) and is committed to leveraging data, people, and smart technology to create a connected ecosystem of solutions that will deliver improved outcomes valued by patients, providers, and payers. A clinical trial of its Smartloop automated insulin delivery system was started in summer 2016 and has subsequently received funding from JDRF. The trial used proprietary closed loop algorithms utilizing Bigfoot’s cloud-connected insulin delivery system and incorporates Dexcom technology in the loop.
“Life with T1D using today’s tools is unreasonable,” says Bigfoot’s founder and CEO Jeffrey Brewer, “Therapies are too expensive, too complex, and require intense focus and attention with little support. Also, healthcare providers are overwhelmed, with too many hurdles blocking them from helping their patients. The Smartloop automated insulin delivery system has the potential to change people’s lives. It is designed to deliver better health with greater ease of use, making life simpler for both the patient and the clinicians who treat them, ultimately lowering cost of treatment.
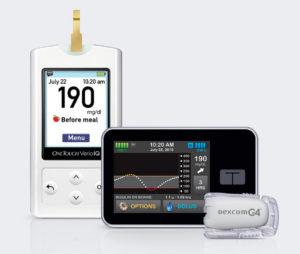
The t-slim insulin pump and Dexcom G4 with Verio iQ meter.
Automated Glucose Control
Insulet Corporation, the company behind the Omnipod tubeless insulin pump technology, announced a year ago (Feb 2016) that it had entered a license agreement and partnership with Mode AGC (Automated Glucose Control), to develop and incorporate the advanced artificial pancreas algorithm created by researchers Francis Doyle PhD, Eyal Dassau PhD, and Howard Zisser MD. This alliance moves forward Insulet’s mission to improve the quality of life for individuals with insulin-dependent diabetes.
Insulet’s artificial pancreas system will use the innovative and differentiated Omnipod platform, the latest Dexcom CGM technology and the algorithm licensed from Mode AGC. The algorithm was create by Drs. Doyle, Dassau and Zisser during their tenure at the University of California, Santa Barbara, and further developed by Mode AGC, a California-based early-stage medical device company founded by prominent diabetes technology specialist Thomas Peyser PhD and diabetes advocate and entrepreneur Jennifer Schneider MD. Insulet has worked with Mode AGC and the inventors to optimize the performance of the algorithm for incorporation into a commercial product. Coupled with the tubeless OmniPod platform, this system should deliver not only improved clinical outcomes, but the greatly desired improvement in quality of life for those living with insulin-dependent diabetes.
See also the page on the ‘bionic’ pancreas being developed at Boston University (p.) and additionally an overview of four AP trails being part funded by the National Institutes for Health in the US (p.).
In our next issue there will be a specific update on the BiAP pump being investigated by the Diabetes Technology Research Group (Imperial College London at St Mary’s Hospital) using a ‘bio-inspired’ approach – mimicking the physiology of the human body, as well as an overview of current – and coming – CGM systems.
Coming soon
This probably all seems like pie-in-the-sky, technology that is talk about but you never get to see. But slowly people in the UK are getting access to insulin pumps and CGM. The latter, while not highly used in the UK, is increasingly being used, though many people are self-funding it if they can’t get it on the NHS. Next to buying an insulin pump, CGM is relatively affordable and can give a lot of insight to someone’s diabetes control, whether you have Type 1 or Type 2. Be prepared though, you get a lot of data and it can be hard to see the wood from the trees, as it were. One thing a sensor will highlight is how sluggish insulin really is. Certainly CGM is core to any AP system.
So, do we want, or do we need a closed loop artificial pancreas? Clearly that is each individual’s decision; assessing the pros and cons will be something only you can do with input and support from your healthcare team.

The iLet ‘bionic’ artificial pancreas.
One of the upsides being promised is that these new AP systems will require little or no old-fashioned finger-prick blood tests. That’s probably a fairly minor upside, how big a deal is finger-pricking? Overtime the pain of finger-pricking wanes next to the pain of having to stop, grab the various bits of kit to do the test, then put it all way again, all without spilling your test strips in the process.
Highs and lows of insulin
A bigger plus for those of us managing the highs and lows of insulin-treated diabetes is that we can realistically expect improved glucose control, meaning lower HbA1c results, and therefore a lower risk of developing long-term complications. Results from trials are all also showing an increased amount of time spend ‘in range’, so not being too high or too low which sounds reassuringly more comfortable that feeling wobbly from a hypo, or feeling headachy and thirsty from being hyper. That would acquaint to improved quality of life, though we should not forget that despite how great this all sounds, we would still have the condition, and we would still have to fight for access to these solutions until such time as they become standard.
The ‘bionic’ pancreas is likely to come out in two forms, a bi-hormonal version and an insulin-only version, both in the form of a iLet device. The second hormone required in the bi-hormonal pump, glycogen, is still undergoing trials, as it’s new and unstable in current formats.
By the time any of these APs become available they will all have apps that mean that you will easily be able to see what’s going on with your diabetes control by looking at the app on your smartphone, so you won’t be out of the loop entirely. And for what it’s worth, I personally believe that nothing beats a blood test if you want to really know what’s going on.
While the human factor will always be involved – they will after all be strapped onto humans – medical advice from those working on these research trials is to ‘let the machine do the work’. It seems that things go wrong when the diabetics on the trials take matters into their own hands and intervene. It seems that, as with most new technology, it will take us a while to learn to trust it, which sounds sensible to me.
One of the downsides will be access – how long will it take before artificial pancreas pumps are really widely available? Although there is a lot of research under way, it seems like we’re a few years away from there being a good choice available. The only one looking to be available this year in the UK is the Mini-Med 670G from Medtronic although there is no date set for that yet.
GLOSSARY
Algorithm: a procedure or formula for solving a problem, based on conducting a sequence of specified actions.
Artificial Pancreas (AP): a closed-loop insulin delivery system using a pump.
Artificial Pancreas Device (APD): another way of referring to an AP.
Automated insulin delivery: insulin dose decisions made by a machine not a person.
Bionic Pancreas: the name used by researchers at Boston University for their bi-hormonal pump.
Bi-hormonal: A closed-loop system using both insulin and glucagon to balance out blood glucose control
Continuous Glucose Monitoring (CGM): CGM sensors take a reading of the glucose level in interstitial body fluid. The results can be seen on a reader or an insulin pump screen in the form of a line on a graph. Recommended for anyone on insulin whether via insulin pump or injection.
Closed Loop insulin delivery: insulin delivery (or suspension) based on CGM readings with no need for the user to do blood tests or adjust doses (some systems may need blood tests for calibration once or twice a day).
Hybrid closed-loop: An insulin pump programmed to both to lower (or even suspend) as well as to increase insulin delivery based on CGM readings.
SAP: Sensor-augmented insulin pump therapy, such as Medtronic’s SmartGuard technology, 640G insulin pump and Enlite CGM allowing for low-glucose suspend by the pump based on low CGM readings.
News items and features like this appear in the Desang Diabetes Magazine, our free-to-receive digital journal. We cover diabetes news, diabetes management equipment (diabetes kit) and news about food suitable for a diabetic diet. Go to the top of this page to sign up – we just need your email address.